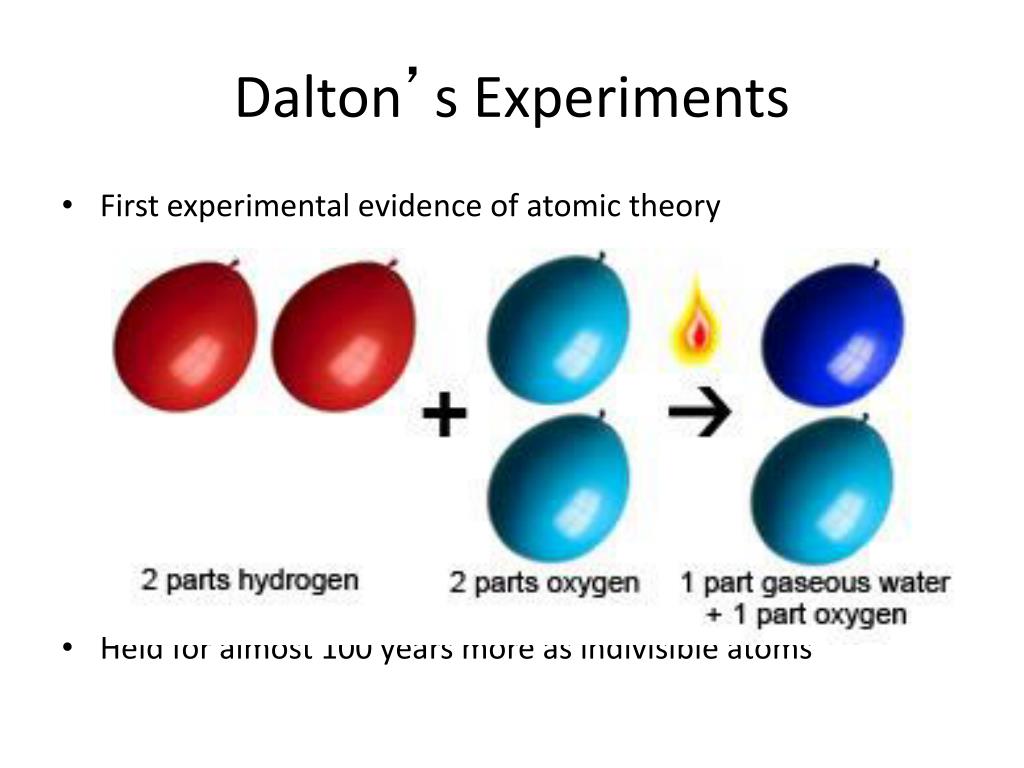
 This caused him to believe that the formula for water was HO instead of H2O and ammonia was NH instead of NH3.ĭalton had made the same mistake that many had before. He believed that when atoms combined in only one ratio, then it needed to be assumed that it would be a binary ratio. This would help to explain why weight ratios in various gases were simple multiples of each other.ĭalton had another postulate that he included with his initial atomic theory that, unfortunately, made it difficult for the scientific community to accept his ideas in their entirety. If an element reacts, their atoms may sometimes combine into more than one simple whole-number ratio.This indicates that when a chemical reaction occurs, it is because the atoms are being rearranged in such a way that they form a different combination. All chemical reactions are a rearrangement of atoms.A combination may also include more than two. 
For a compound to form, Dalton suggested with his atomic theory that it would have to be composed of at least two different types of atoms.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
